How Non-Contact Infrared Thermometers Are Transforming Home Healthcare and Infection Control in 2026

Non-contact infrared thermometers have evolved from a niche clinical tool into an essential household device found in millions of homes, schools, and workplaces around the world. As we move deeper into 2026, the global infrared thermometer market continues to expand at a remarkable pace, driven by heightened infection control awareness, growing consumer demand for fast and hygienic temperature screening, and a new generation of smarter, more accurate devices entering the market.
For healthcare distributors, OEM buyers, and retail brands, the infrared thermometer category represents one of the most consistent and profitable segments in the home medical device industry. Here is a comprehensive look at the technology, market dynamics, and sourcing considerations that are shaping this sector in 2026.
The Science Behind Non-Contact Temperature Measurement
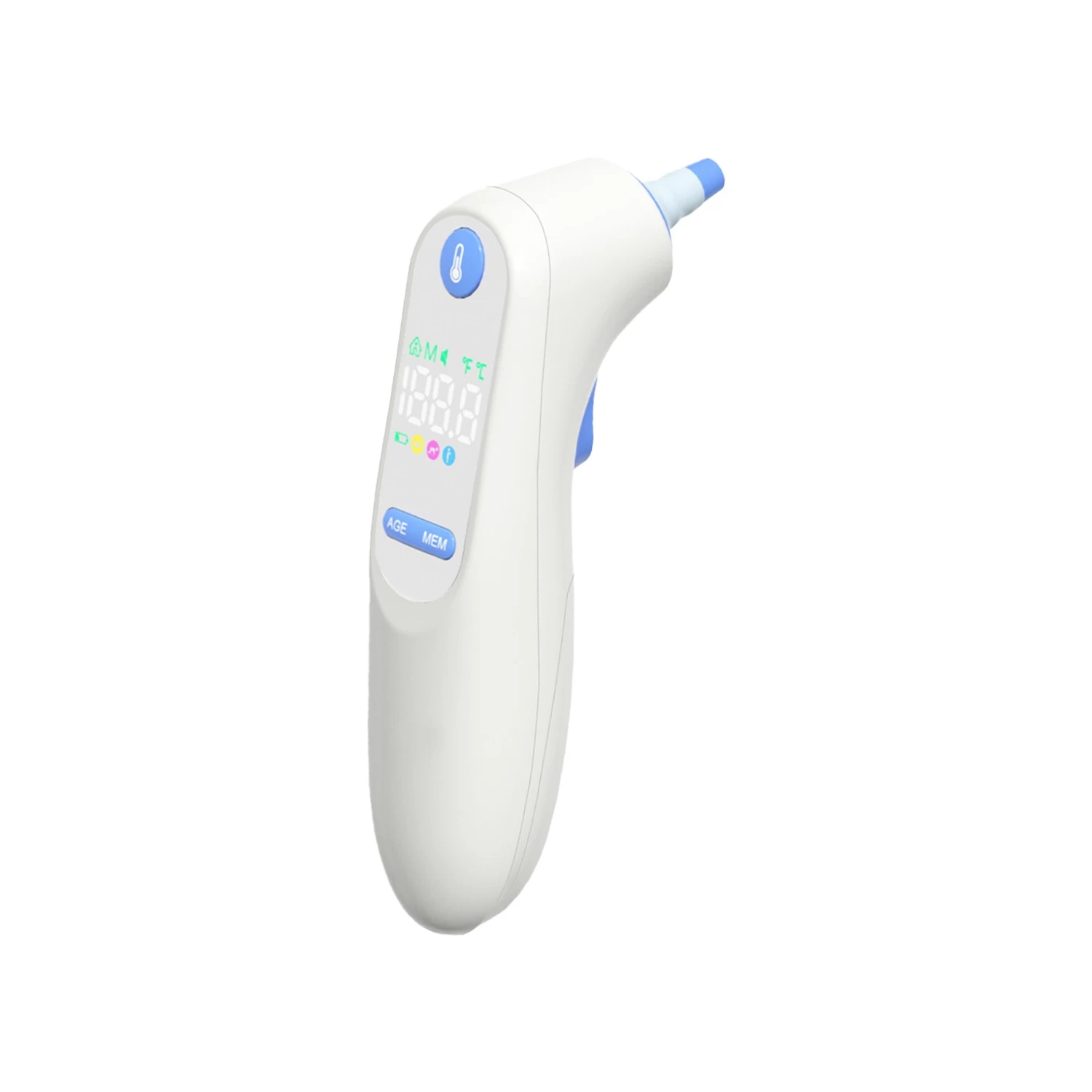
Non-contact infrared forehead thermometers work by detecting the thermal radiation emitted by the human body, converting that energy into a temperature reading displayed on an LCD screen in as little as one second. Unlike traditional mercury or digital contact thermometers, infrared devices require no physical contact with the skin, making them inherently more hygienic and significantly faster to use.
This contactless measurement capability has proven invaluable in settings where cross-contamination is a concern. Hospitals, clinics, airports, schools, factories, and retail establishments have all adopted non-contact infrared thermometers as a standard screening tool. At home, parents rely on these devices to check their children's temperature without disturbing a sleeping child, while caregivers use them for quick daily monitoring of elderly family members.
Modern infrared thermometers have addressed many of the accuracy concerns that plagued earlier generations. Advanced sensor technology, combined with sophisticated algorithms that account for ambient temperature variations and skin emissivity differences, now delivers clinical-grade accuracy that meets FDA and CE MDR standards. The best devices on the market achieve measurement precision within plus or minus 0.2 degrees Celsius, putting them on par with traditional clinical thermometers.
Market Growth That Shows No Signs of Slowing
The global infrared thermometer market is projected to reach $3.8 billion by 2026, expanding at a 7.9% compound annual growth rate. The handheld segment alone is growing at an impressive 12.4% annual rate through 2033, reflecting sustained consumer and institutional demand.
Asia Pacific continues to lead in adoption volume, driven by large populations, expanding healthcare infrastructure, and strong manufacturing capabilities in countries like China. North America delivers the highest profitability per unit, while European growth is being fueled by healthcare system modernization programs and stricter workplace safety regulations.
Several factors are sustaining this growth trajectory beyond the initial pandemic-driven surge. Permanent changes in infection control protocols across healthcare facilities and workplaces have created ongoing institutional demand. The expansion of telemedicine and remote patient monitoring programs has made home temperature tracking a routine part of chronic disease management. And rising consumer health awareness, particularly among young parents, has established infrared thermometers as a must-have item in every family medicine cabinet.

Key Features Driving Consumer and Buyer Preferences
The infrared thermometer market in 2026 is increasingly defined by feature differentiation rather than price competition alone. Buyers and consumers are looking for specific capabilities that enhance accuracy, usability, and value:
Fever Alert Systems with Color-Coded Displays: Modern infrared thermometers use green, orange, and red backlight indicators to instantly communicate whether a reading is normal, elevated, or indicates fever. This intuitive visual feedback eliminates the need for users to memorize temperature thresholds and is particularly valued by parents and elderly users.
Age-Mode Calibration: Because normal body temperature ranges differ between infants, children, and adults, leading thermometers now include age-mode settings that automatically adjust fever alert thresholds. This feature has become a key differentiator for products targeting the family and pediatric care market.
Dual-Mode Measurement: Many premium infrared thermometers offer both forehead (body) and surface (object) measurement modes, allowing users to check not only body temperature but also the temperature of baby bottles, bathwater, food, and room environments. This versatility increases the perceived value and daily utility of the device.
Memory Storage and Data Tracking: Devices with 30 to 50 reading memory capacity allow users to track temperature trends over time, which is particularly important for monitoring the progression of illness or the effectiveness of fever-reducing medications.
Silent Mode and Backlit Displays: Features designed for nighttime use, such as muted measurement beeps and bright backlit screens, are highly valued by parents who need to check a sleeping child's temperature without waking them.
Compact Ergonomic Design: The trend toward smaller, lighter devices with intuitive one-button operation continues to accelerate. Devices measuring under 150mm in length and weighing less than 100 grams are now standard, making them easy to store in a drawer, carry in a handbag, or keep in a car glove compartment.
Regulatory Compliance: The Foundation of Market Access
For OEM buyers and private label brands sourcing infrared thermometers, regulatory compliance is not optional - it is the prerequisite for market entry. The regulatory landscape in 2026 continues to tighten, with authorities worldwide demanding higher standards of accuracy and safety.
FDA clearance remains essential for the US market, where non-contact infrared thermometers are classified as Class II medical devices. The European market requires CE MDR certification under the new Medical Device Regulation framework, which imposes stricter clinical evidence and post-market surveillance requirements than the previous MDD directive.
TGA approval is mandatory for Australia, while markets across Southeast Asia, the Middle East, and Latin America increasingly require ISO 13485 quality management system certification as a minimum standard. MDSAP certification, which satisfies the regulatory requirements of the US, Canada, Australia, Brazil, and Japan through a single audit, is becoming the gold standard for manufacturers seeking efficient multi-market access.
Beyond product-level certifications, social compliance credentials such as BSCI are increasingly important for buyers who must demonstrate ethical sourcing practices to their customers and regulatory authorities, particularly in European markets.
Choosing the Right Manufacturing Partner
The infrared thermometer market is highly competitive, with hundreds of manufacturers offering seemingly similar products at varying price points. For buyers looking to build a successful product line, the choice of manufacturing partner can make or break their market entry.
Key criteria for evaluating potential suppliers include: proven production scale with over a decade of experience in medical device manufacturing, a comprehensive portfolio of regulatory certifications covering target markets, demonstrated market success through rankings on major e-commerce platforms like Amazon and B2B platforms like Alibaba, flexible OEM and ODM capabilities for custom branding and feature configurations, and reliable quality management systems backed by ISO 13485 certification.
The strongest manufacturers are those that combine all of these elements while also offering competitive pricing through vertically integrated production, dedicated R&D teams that continuously develop new features and models, and experienced export teams that understand the documentation and logistics requirements of different markets.
The Road Ahead: What to Expect in the Coming Years
The infrared thermometer category is far from mature. Several emerging trends will shape the next phase of market development. Smart connectivity features, including Bluetooth integration with mobile health apps, will transform standalone thermometers into connected health monitoring devices that feed data directly into personal health records. AI-powered algorithms will further improve accuracy by learning individual user patterns and environmental factors. And the convergence of infrared thermometry with other vital sign measurements may eventually produce compact multi-function devices that measure temperature, heart rate, and blood oxygen in a single scan.
For businesses positioned in the home medical device supply chain, the infrared thermometer segment offers a compelling combination of strong margins, consistent demand, growing market size, and clear product differentiation opportunities. The key to success lies in partnering with the right manufacturer and bringing genuinely innovative, compliant products to market ahead of the competition.