Digital Ear Thermometers and Blood Pressure Monitors: How Smart Factory Innovation Is Reshaping Home Medical Device Manufacturing in 2026

The home medical device industry is experiencing a manufacturing revolution. As consumer demand for digital ear thermometers and digital blood pressure monitors continues to accelerate globally, the factories behind these products are investing heavily in smart production systems, automated quality control, and flexible OEM capabilities that allow them to serve diverse international markets from a single production base.
For distributors, importers, and brand owners sourcing digital ear thermometers or digital blood pressure monitors, understanding the factory landscape in 2026 is just as important as understanding the end consumer. The quality of your manufacturing partner directly determines the accuracy, reliability, and regulatory compliance of every device that reaches your customer's hands.
Why Digital Ear Thermometers Are Gaining Market Share
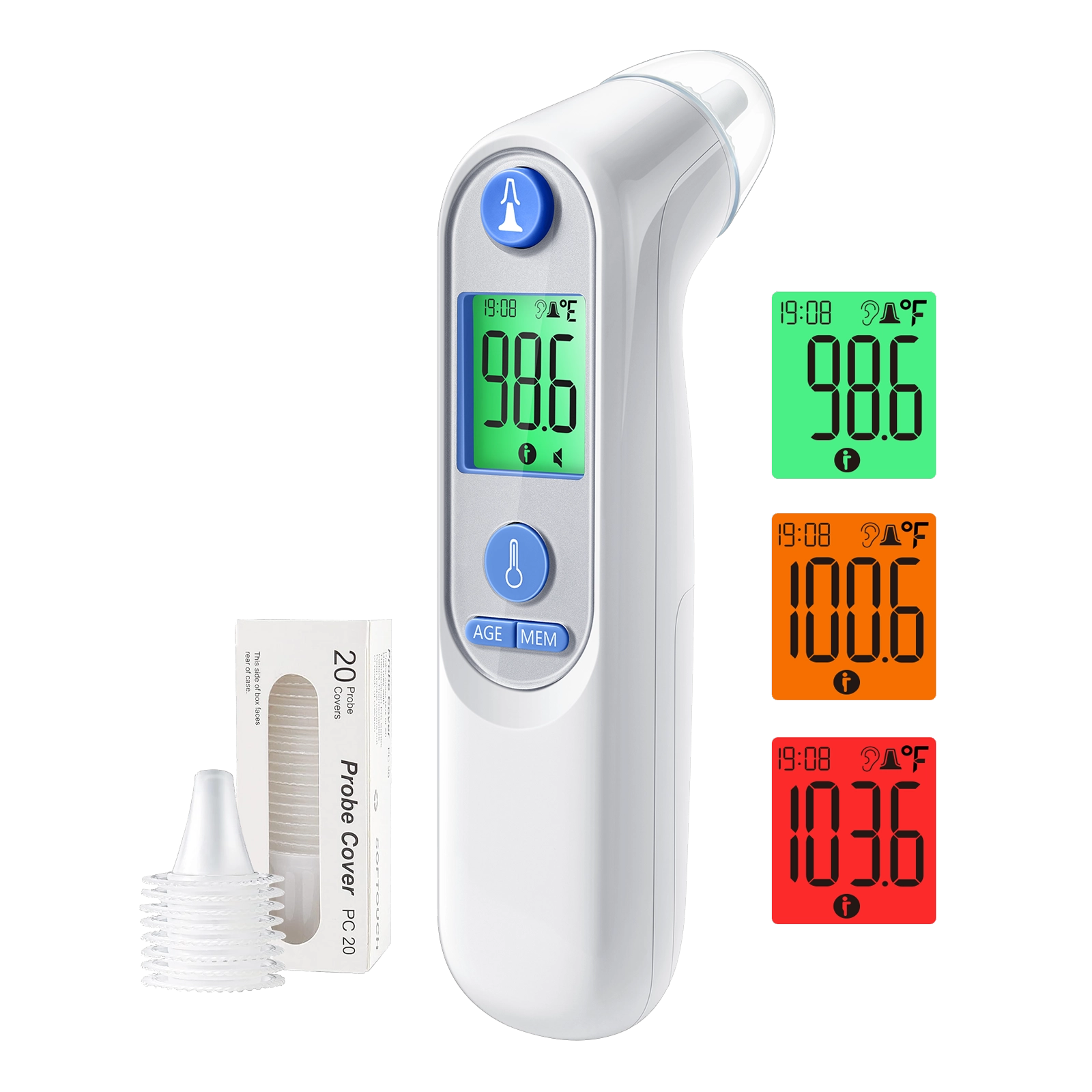
Digital ear thermometers have carved out a distinct and growing segment within the broader thermometer market. Unlike forehead infrared thermometers that measure surface skin temperature, a digital ear thermometer reads the infrared heat generated by the eardrum and surrounding tissue, providing a measurement that closely reflects the body's core temperature.
The global baby ear thermometer market alone is growing at a 9.6% compound annual growth rate, reflecting strong demand from parents seeking fast, accurate, and gentle temperature measurement for infants and young children. The household segment for digital ear thermometers is expanding at an even faster pace, driven by consumer recognition that ear measurement delivers consistently reliable readings with minimal technique sensitivity.
Clinical studies have repeatedly demonstrated that tympanic (ear) temperature measurement correlates strongly with core body temperature, making the digital ear thermometer a preferred choice in many pediatric clinics, hospitals, and home care settings. Modern digital ear thermometers achieve accuracy within plus or minus 0.2 degrees Celsius when used according to manufacturer instructions, meeting the rigorous standards set by ASTM International and ISO 80601-2-56.
Key features driving consumer preference for digital ear thermometers in 2026 include one-second measurement speed, color-coded fever alert displays, memory storage for up to 20 previous readings, age-mode calibration for infants and adults, and disposable probe covers that ensure hygienic use across multiple family members.
The Digital Blood Pressure Monitor Factory: What Has Changed
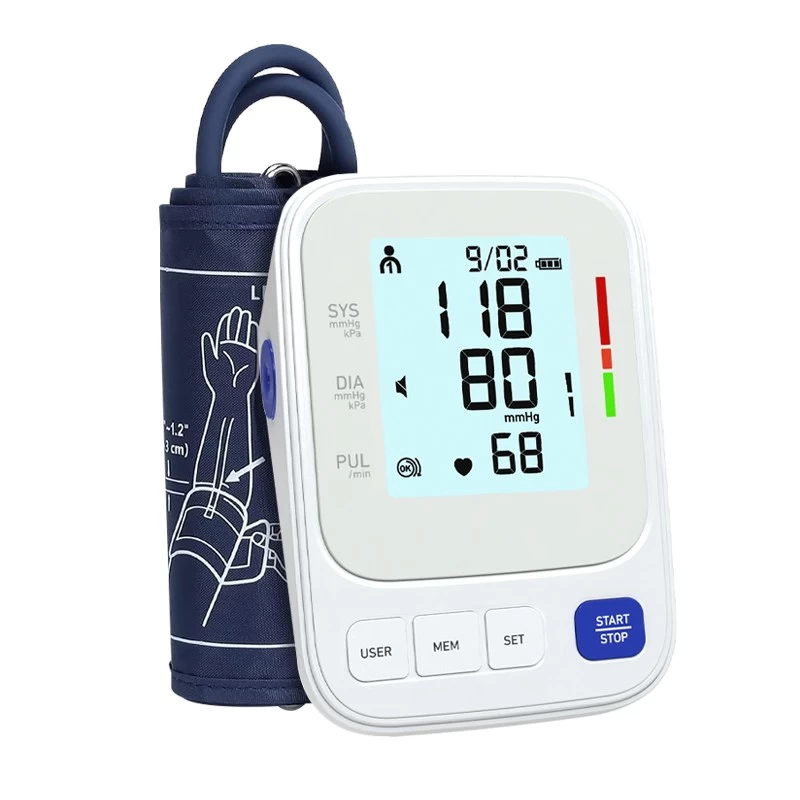
The digital blood pressure monitor market continues its upward trajectory, with the global market valued at $2.27 billion in 2025 and projected to reach $5.02 billion by 2035 at an 8.26% CAGR. Behind these numbers lies a dramatic transformation in how digital blood pressure monitors are manufactured.
Leading digital blood pressure monitor factories in 2026 operate with Industry 4.0 principles at their core. Automated surface mount technology lines place thousands of electronic components per hour with micron-level precision. Computer vision inspection systems check every solder joint, component placement, and assembly step in real time, catching defects that human inspectors might miss. Environmental testing chambers simulate years of use in hours, ensuring that every digital blood pressure monitor leaving the factory meets its rated lifespan specifications.
The most advanced digital blood pressure monitor factories maintain vertically integrated production, controlling everything from PCB fabrication and sensor calibration to cuff manufacturing, firmware programming, and final packaging. This vertical integration delivers three critical advantages: tighter quality control at every production stage, faster response to custom OEM requirements, and lower costs through elimination of intermediary markups.
Production capacity matters enormously in the digital blood pressure monitor business. Seasonal demand spikes, particularly during health awareness campaigns and flu seasons, can strain unprepared suppliers. Factories with capacity to produce hundreds of thousands of units per month while maintaining consistent quality are the ones that earn long-term partnerships with major international brands.
Smart Manufacturing Technologies Driving Quality
Both digital ear thermometer and digital blood pressure monitor production benefit from the same smart manufacturing technologies that are transforming factory floors worldwide.
AI-Powered Quality Inspection: Machine learning algorithms trained on millions of product images can detect cosmetic defects, misaligned components, and assembly errors with accuracy exceeding 99.5%. These systems operate continuously without fatigue, ensuring consistent inspection quality across all shifts.
Automated Calibration Systems: Every digital ear thermometer must be individually calibrated against a precision blackbody radiation source to ensure measurement accuracy. Similarly, every digital blood pressure monitor undergoes automated calibration against reference pressure standards. Modern factories use robotic calibration stations that process hundreds of units per hour while maintaining traceability records for every individual device.
Real-Time Production Monitoring: Factory management systems collect data from every machine, sensor, and test station on the production line, presenting real-time dashboards that track output rates, defect rates, and equipment performance. This data-driven approach enables predictive maintenance that prevents unexpected downtime and production disruptions.
Clean Room Assembly: Medical device manufacturing requires controlled environments to prevent contamination. Digital ear thermometer sensor assemblies are particularly sensitive to dust and particulates. Leading factories maintain ISO Class 7 or Class 8 clean rooms for critical assembly steps.
Full Traceability Systems: Regulatory authorities increasingly require manufacturers to demonstrate complete traceability from raw materials through finished products. Modern factories assign unique identifiers to every unit, linking each device to its component lot numbers, calibration data, test results, and production personnel.

Regulatory Compliance Built Into the Factory
For buyers sourcing digital ear thermometers or digital blood pressure monitors, regulatory compliance is not an afterthought applied at the end of the production process. It must be embedded throughout the factory's quality management system.
ISO 13485 certification is the foundational requirement, establishing a quality management framework specifically designed for medical device manufacturing. This standard governs everything from design controls and risk management to purchasing controls, production processes, and corrective action procedures.
FDA registration and 510(k) clearance are mandatory for selling digital ear thermometers and digital blood pressure monitors in the United States market. The FDA classifies both product types as Class II medical devices, requiring manufacturers to demonstrate substantial equivalence to predicate devices and maintain compliance with current Good Manufacturing Practice regulations.
CE MDR certification under the European Union's Medical Device Regulation requires even more extensive documentation, including clinical evaluation reports, post-market surveillance plans, and technical files that demonstrate conformity with essential safety and performance requirements.
MDSAP certification allows a single audit to satisfy the regulatory requirements of five countries simultaneously: the United States, Canada, Australia, Brazil, and Japan. For digital blood pressure monitor and digital ear thermometer factories serving global markets, MDSAP certification streamlines the compliance burden while demonstrating commitment to the highest quality standards.
Beyond product certifications, social compliance audits such as BSCI are increasingly required by European buyers who must demonstrate ethical sourcing throughout their supply chains.
What OEM Buyers Should Evaluate in a Factory Partner
Selecting the right factory for digital ear thermometers or digital blood pressure monitors requires evaluation across multiple dimensions that go beyond quoted unit prices.
Technical Capabilities: Does the factory have dedicated R&D teams that can modify existing designs or develop entirely new products to your specifications? Can they handle firmware customization for different languages, measurement units, and market-specific features?
Regulatory Portfolio: Does the factory already hold FDA, CE MDR, ISO 13485, and MDSAP certifications for the product categories you need? Obtaining these certifications from scratch can take 12 to 18 months, so partnering with a pre-certified factory dramatically accelerates your time to market.
Production Flexibility: Can the factory handle both large volume orders for established products and smaller initial runs for new market entries? The ability to scale production up or down based on demand without minimum order quantities in the hundreds of thousands is valuable for growing brands.
Reference Clients: Does the factory supply recognized brands that you can verify? A track record of successful partnerships with brands sold on Amazon, in pharmacies, or through hospital procurement channels provides confidence in consistent quality delivery.
Export Experience: Does the factory have experienced export teams that understand the documentation, labeling, and logistics requirements for your target markets? International shipping compliance, customs documentation, and market-specific packaging requirements can create costly delays if the factory lacks export expertise.
Emerging Trends Shaping the Next Generation
The digital ear thermometer and digital blood pressure monitor categories are both evolving rapidly, and the factories that invest in next-generation capabilities today will lead the market tomorrow.
Connected Health Integration: Both digital ear thermometers and digital blood pressure monitors are increasingly expected to feature Bluetooth connectivity that syncs measurements with smartphone health applications. Factories must integrate wireless communication modules and develop companion app firmware alongside the core medical device hardware.
Multi-Function Devices: The convergence trend is creating demand for devices that combine multiple measurement capabilities. Combination forehead and ear thermometers are already common, and the next generation may integrate pulse oximetry or heart rate measurement into a single handheld device.
Sustainable Manufacturing: Environmental regulations and consumer expectations are pushing factories toward recyclable packaging materials, reduced energy consumption in production, lead-free soldering processes, and product designs that minimize electronic waste.
AI-Enhanced Accuracy: Machine learning algorithms that improve measurement accuracy by accounting for environmental conditions, user technique variations, and individual physiological differences represent the next frontier in device intelligence.
Building a Competitive Advantage Through Factory Partnership
In the home medical device market of 2026, product differentiation increasingly comes from manufacturing excellence rather than feature lists alone. The digital ear thermometer or digital blood pressure monitor that reaches the consumer with superior accuracy, reliability, and build quality wins market share regardless of whether it carries a premium or value price tag.
For businesses seeking to enter or expand their presence in the digital ear thermometer or digital blood pressure monitor markets, the manufacturing partner you choose is your most important strategic decision. A factory with proven regulatory credentials, flexible OEM capabilities, smart manufacturing systems, and deep export experience provides the foundation upon which successful product lines are built.
The opportunity is substantial. The technology is mature yet still innovating. And the consumers are waiting for accurate, connected, and well-built devices that help them manage their health at home with confidence.
Keywords: digital ear thermometer, digital blood pressure monitor, ear thermometer factory, blood pressure monitor OEM manufacturer, digital ear thermometer accuracy, FDA certified ear thermometer, CE MDR blood pressure monitor, smart medical device factory, home medical device manufacturing 2026, OEM ear thermometer supplier